Clinical trials begin for promising cancer treatment discovered at Southampton
A new type of cancer treatment developed at Southampton – that it is hoped will significantly improve the success rate of immunotherapy – is undergoing clinical trials for the first time.
Immunotherapy, which helps the immune system recognise and attack cancer cells, has been the biggest breakthrough in cancer treatment in recent years. However, it is only effective in about 20% of patients.
Research led by the University of Southampton, supported by Cancer Research UK, has shown that a new drug – originally developed to treat organ tissue scarring, or fibrosis – could significantly improve the success rate of immunotherapy.
Gareth Thomas, Professor of experimental pathology at Southampton, has been working on the research for about ten years. He explained: “Our research has been focused on a type of normal cell found in cancers – cancer-associated fibroblasts (CAFs). Cancers containing lots of these normal cells tend to behave aggressively and are resistant to treatment.
Breaking down tumours’ shields
“We found that CAFs shield tumours from the patient’s immune system, preventing ‘killer’ lymphocytes from penetrating the tumour to combat malignant cells; this is significant in terms of immunotherapy, which works by boosting this immune response. We also identified an enzyme called NOX4, which is important in the formation of CAFs. We found that inhibiting NOX4 prevented CAF formation and improved response to immunotherapy.”
Professor Thomas and his team, through the University of Southampton, have now signed an Intellectual Property (IP) Agreement with Calliditas Therapeutics, a Swedish pharmaceutical company, which has developed a NOX4 inhibitor that is currently being tested to treat kidney, liver and lung fibrosis.
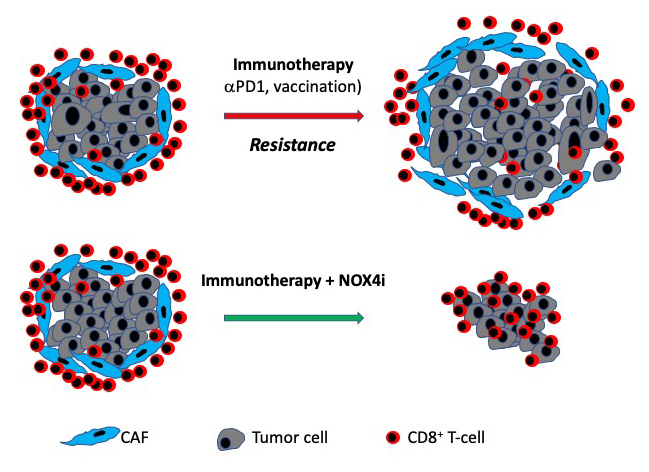
The clinical trial, underway at over 30 centres in Europe and United States, is testing the effectiveness of this NOX4 inhibitor (setanaxib) in combination with pembrolizumab, one of the most common immunotherapy drugs.
Resistance mechanism
“Even though immunotherapy has produced very exciting results, most patients fail to respond,” said Professor Thomas. “The presence of CAFs is recognised as a resistance mechanism, and lots of patients have this type of cell in their cancers. This new treatment could potentially be of great benefit to many patients. It will be really exciting to see the outcomes of the clinical trial.”
The phase 2 clinical trial is enrolling patients with recurrent or metastatic head and neck cancer, around 80% of whom have CAF-rich tumours. The trial will enrol approximately 50 patients at sites in Europe and the USA, which in the UK include Poole Hospital, The Royal Marsden Hospital, The Royal Liverpool University Hospital and St Bartholomew’s Hospital.
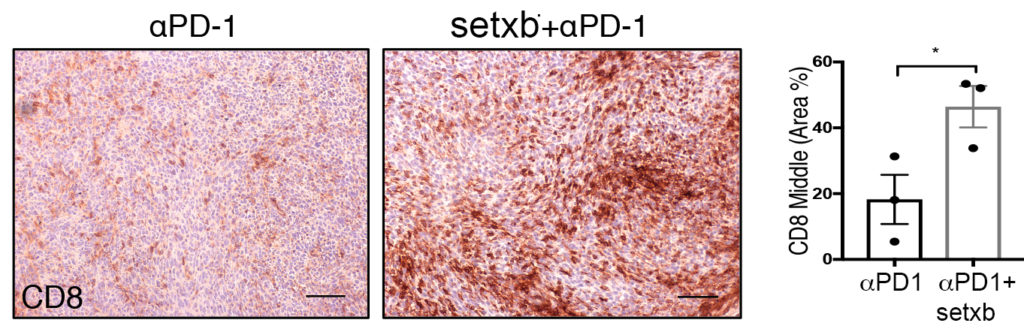
“We are very excited to have initiated this phase 2 clinical trial of setanaxib in patients with recurrent or metastatic head and neck cancer,” said Dr Richard Philipson, chief medical officer of Calliditas Therapeutics. “This marks an important step in our long-term collaboration with Professor Thomas and his team at the University of Southampton. We look forward to continuing the collaboration and building further on the work that Professor Thomas continues to do in the field of cancer-associated fibroblasts.”
Project contact

Professor Gareth Thomas
Professor of Experimental Pathology, University of Southampton

